…Thou shalt take of the first of all the fruit of the ground, which thou shalt bring in from thy land that the Lord thy G‑d giveth thee; and thou shalt put it in a basket and shalt go unto the place which the Lord thy G‑d shall choose to cause His name to dwell there. And thou shalt come unto the priest that shall be in those days, and say unto him: ‘I profess this day unto the Lord thy G‑d, that I am come unto the land which the Lord swore unto our fathers to give us.’ And the priest shall take the basket out of thy hand, and set it down before the altar of the Lord thy G‑d.
Deuteronomy 26:2-4
The debate about the nature of matter goes back to antiquity. A pre-Socratic Greek philosopher Leucippus (Fl. 5th c. BCE), and his student, Democritus (c. 460 –c. 370 BCE) are considered to be the fathers of the atomic theory of nature. They held that everything consists of indivisible atoms. Their successors Epicurus (341–270 BCE) further developed this idea. Roman philosopher Lucretius (99 BCE – c. 55 BCE) presented a very lucid argument from decay in support of atomism in his poem, De Rerum Natura (On the Nature of Things).
At the beginning of the 11th Century, the Arabic scientist Alhazen developed the first comprehensive theory of optics based on the notion that the rays of light were composed of particles of light. In 1670, Isaac Newton formulated the corpuscular theory of light (corpusculum in Latin means a small body or a particle). In the 19th century, a number of scientists including Antoine Lavoisier, Joseph Louis Proust, John Dalton, Amedeo Avogadro, and Dimitri Mendeleev contributed to the development of the atomic theory and its applications to physics and chemistry. In his famous 1905 paper on Brownian motion, Einstein finally provided firm theoretical support for the atomic theory.
Using the atomic theory of the black body (an ideal body that absorbs 100% of light without reflecting any of it back), in 1900, Max Planck proposed that the energy (E) light emitted by the black body is proportionate to the frequency (v) of the oscillator that emitted it:
E = hν,
where E is the energy, v is the frequency, and h is the Planck constant. In another landmark 1905 paper, Einstein explained the photoelectric effect by postulating that energy is emitted and absorbed in discrete quantities—quanta.
Quantum revolution was born. In 1921, Einstein was awarded a Nobel Prize for the theory of photoelectric effect.
Parallel with the corpuscular theory of light, a competing wave theory was being developed. In 1630, René Descartes proposed a wave theory of light. He conceived of light as wave-like disturbances in a universal medium (“plenum” later replaced by aether). Newton’s contemporaries Robert Hooke and Christiaan Huygens (and later Augustin-Jean Fresnel) further developed the wave theory of light. Thomas Young’s 1803 double-slit experiment producing interference seemed to have confirmed the wave theory of light.
Thomas Young’s sketch of two-slit diffraction of waves, 1803
At the end of the 19th century, James Clerk Maxwell applied his theory of electromagnetism to explain light waves. He demonstrated that visible light, ultraviolet light, and infrared light were all electromagnetic waves of differing frequencies. So what is light – particles or waves?
In 1924, French physicist, Louis-Victor de Broglie, hypothesized that not just light but all matter has a wave-like nature:
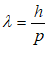

Where λ is wavelength, p is momentum and h is Planck constant. This formula is a simple generalization of Einstein’s formula used to explain the photoelectric effect. The wave nature of electrons was confirmed three years later in experiments showing the diffraction of electrons. De Broglie was awarded a Nobel Prize for his discovery of the wave nature of particles in 1929. Later, the wave behavior of other particles, such as protons and neutrons, and even atoms and molecules was also observed.
So, is matter made up of particles (as ancient Greek atomists thought) or waves? Niels Bohr thought that “particle” and “wave” were classical concepts, which did not fully describe reality on a microscopic level. He proposed his principle of complementarity postulating that these two competing views of reality complement each other. A quantum-mechanical object is neither a particle nor a wave. Sometimes, when it has high energy and, therefore, short de Broglie wavelength, it behaves more like a particle. In other cases, when the energy is low, and de Broglie wavelength is substantial, it behaves more like a wave.
The debate about the wave-particle dualism of nature rages to this day. Nobel Laureate Richard Feynman, who significantly contributed to the development of quantum field theory, conceived of fields as clouds of virtual particles exchanging energy between particles of matter. In this view, the electromagnetic field is a collection of virtual photons carrying energy between electrons. Another Nobel Laureate, Julian Schwinger who contributed greatly to the development of quantum electrodynamics, perceived particles as wave pockets. The pilot wave theory developed by de Broglie and Bohm maintains wave-particle dualism—a quantum particle is guided by a pilot wave (see my earlier post “The Pilot Wave“).
Perhaps this Torah portion, Ki Tavo, hints at the wave-particle dualism. Bikurim—the first fruits offered as a sacrifice—represent the particle view of the world. Fruits are separate and countable. However, they are placed together in a basket. In Torah exegesis, we have dialectic tension between prat and klal. Countable items, like fruits, represet prat (particular), whereas a collection as a whole represents klal (“general” or “collective” or “whole”). Prat-klal duality is, therefore, parallel particle-wave duality of matter.
Furthermore, commeting on the verse:
And the priest shall take the basket out of thy hand: in order to wave it.
Deuteronomy 26:4
Rashi explains that the priest waved the basket in front of the altar. This appears to be hinting at the wave view. Thus, a fruit basket waved in front of the altar can be viewed as a metaphor for particle-wave duality.
Particles or waves, we elevate and unify both views by offering Bikurim at the alter emphasizing their common source:
I have brought the first of the fruit of the land, which Thou, O Lord, hast given me.
Deuteronomy 26:10

The offering of the First Fruits — Bikkurim
Illustration from a Bible card published between 1896 and 1913 by the Providence

The analogy is is a bit of a reach, but not ridiculously so. Indeed, there is the indivisibility of the individual components of fruit. There are properties attached to the borh individual and collective identities. Once waved another characteristic emerges, as a function of the wave. Charming, clever and informative drosha!